Thermodynamics - Online Test
Q1. Spontaneity in the context of chemical thermodynamics means
Answer : Option C
Explaination / Solution:
spontaneity means a reaction occurring on its own without any help of external agency.
Q2. Entropy is a state function and measures
Answer : Option A
Explaination / Solution:
Entropy is a state function and it is measure of randomness of a system.
Q3. We can calculate the change in entropy of a reversible process by
Answer : Option B
Explaination / Solution:
is used to measure entropy system undergoin reversible
Q4. If change in Gibbs energy ΔG is negative (< 0) at constant pressure and temperature
Answer : Option B
Explaination / Solution:
ΔG gives a criteria for spontaneous and non spontaneous process at constant temperature and pressure. ΔG < 0 (negative) means reaction is spontaneous and ΔG >0 (positive) means reaction is non spontanenous.
Q5. Standard Gibbs energy change is related to the equilibrium constant of the reaction as follows:
Answer : Option D
Explaination / Solution:
= -RTln K
where is standard Gibbs energy change, K is equilibrium constant, R is universal gas constant and T is temperature in kelvin scale
Q6. A thermodynamic state function is a physical quantity
Answer : Option B
Explaination / Solution:
State function is one which is only dependent on initial and final state of the system and is independent of the path by which that change has occurred.
Q7. For an adiabatic change
Answer : Option B
Explaination / Solution:
An adiabatic process would not allow exchange of heat between the system and surroundings, hence q=0.
Q8. The enthalpies of all elements in their standard states are:
Answer : Option C
Explaination / Solution:
The enthalpies of formation of all elements in their standard states is taken as zero.
Q9. of combustion of methane is - X kJ . The value of is
Answer : Option A
Explaination / Solution:
CH4 (g) + 2O2 (g) → CO2 (g) + 2H2O (l)
We know, RT
Δng is negative as product water in combustion reaction is in liquid state.
This implies ΔHo will be more negative than ΔUo
Hence, ΔHo < ΔUo
Q10. The enthalpy of combustion of methane, graphite and dihydrogen at 298 K are, - 890.3 kJ mol-1, -393.5 kJ mol-1 and –285.8 kJ mol-1 respectively. Enthalpy of formation of CH4 (g) will be
Answer : Option C
Explaination / Solution:
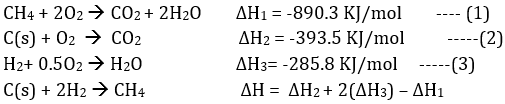
ΔH= -393.5 +2(-285.8)-(-890.3)
= -74.8 kJ/mo
