Thermodynamics - Online Test
A 1.0-kg bar of copper is heated at atmospheric pressure. If its temperature increases from 20C to 50C, what is the work done by the copper on the surrounding atmosphere? Given that for Cu = 5.1 x 10-5 / C,
A 3.00-g copper penny at 25.0C drops from a height of 50.0 m to the ground. (a) If 60.0percent of the change in potential energy goes into increasing the internal energy, what is its final temperature? Specific heat of Cu
Heat used in increasing temp
An ideal gas is enclosed in a cylinder with a movable piston on top. The piston has a mass of 8 000 g and an area of 5.00 and is free to slide up and down, keeping the pressure of the gas constant. How much work is done as the temperature of 0.200 mol of the gas is raised from 20.0 C to 300C?
in isobaric process
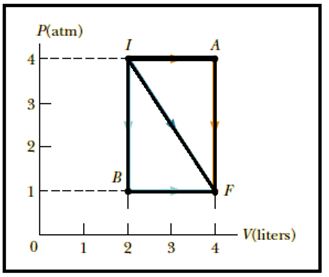
Consider the cyclic process depicted in Figure If Q is negative for the process BC, and if is negative for the process CA, what are the signs of W and that are associated with BC?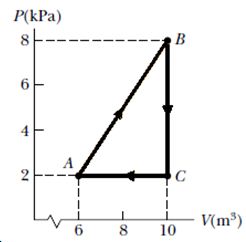
W = area under P-V curve = 0
Consider the cyclic process depicted in Figure if is negative for the process CA, what are the signs of Q and W are associated with CA?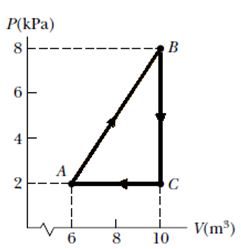
Consider the cyclic process depicted in Figure If Q is negative for the process BC, and if is negative for the process CA, what are the signs of Q, W, and that are associated with AB?

W = +ve (Expansion)
