States of Matter - Online Test
Q1.
Critical temperature of carbon dioxide is the highest temperature at which liquid carbon dioxide is observed. Above this temperature
Answer : Option C
Explaination / Solution:
Tc is the highest Temp where a gas can exist in liquid state. Above this no liquid state exist. It is impossible to compress the gas into liquid above critical temperature, no matter what ever amount of pressure is applied.
Q2. Above the Critical temperature of carbon dioxide
Answer : Option D
Explaination / Solution:
Tc is the highest Temp where a gas can exist in liquid state. Above this no liquid state exist hence it is not possible to liquify the gas no matter what pressure is applied.
Q3. The three states of matter of are in equilibrium at
Answer : Option A
Explaination / Solution:
Triple point is the point where all the three states coexist in equilibrium.
Q4. Surface tension decreases as the temperature is raised because
Answer : Option D
Explaination / Solution:
Surface tension decreases as the temperature is raised because Increase in temperature increases the kinetic energy of the molecules and effectiveness of intermolecular attraction decreases.
Q5. Viscous force is
Answer : Option A
Explaination / Solution:
Viscous force is directly proportional to area and velocity gradient.
Q6. Water has high surface tension and high capillarity because of
Answer : Option D
Explaination / Solution:
Water molecules form extensive Hydrogen Bonds.
Q7. What will be the minimum pressure required to compress 500 of air at 1 bar to 200 at
Answer : Option A
Explaination / Solution:
Using ideal gas equation, PV=nRT,
since T is constant, we have
Q8.
At, the density of a certain oxide of a gas at 2 bar is same as that of dinitrogen at 5 bar. What is the molecular mass of the oxide?
Answer : Option C
Explaination / Solution:
PV=(m/M)RT
Q9. Pressure of 1 g of an ideal gas A at 27 ∘C is found to be 2 bar. When 2 g of another ideal gas B is introduced in the same flask at same temperature the pressure becomes 3 bar. Relationship between their molecular masses is
Answer : Option C
Explaination / Solution:
PV=(m/M)RT
Q10.
The drain cleaner, Drainex contains small bits of aluminum which react with caustic soda to produce dihydrogen. What volume of dihydrogen at and one bar will be released when 0.15g of aluminum reacts?
Answer : Option B
Explaination / Solution:
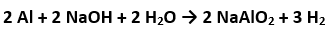
Moles of Al= 0.15/27 =0.0055 moles And 2 mole of Al produces 3 moles of so 0.0055 moles will produce 3/2x0.0055 moles of =0.00825 moles. Now PV=nRT where n=0.00825 , P=1 bar and T=293K.
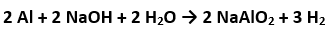
Moles of Al= 0.15/27 =0.0055 moles And 2 mole of Al produces 3 moles of so 0.0055 moles will produce 3/2x0.0055 moles of =0.00825 moles. Now PV=nRT where n=0.00825 , P=1 bar and T=293K.
