Some Basic Concepts of Chemistry - Online Test
Calculations:
Mass of 1mole of O2
= the mass of 6.022 x 1023 molecules of O2 ' ( as per Avogadro's law )
= molar mass of O2 in grams ( ie. gram molar mss )
= 32 .0 g
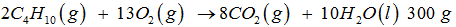
of is combusted in 1000 g of O2. The yield of and the limiting agent in the reaction are
How many molecules of H2O are there in 18g of water?
(Hint: Avagadro’s Number = 6.02 x 1023 atoms/mol)
Since , the number of molecules contained in one mole of a substance
= Avogadro's number (N)
= 6.022 x 1023 molecules ,
and , gram molar mass of water
= 18 g
18g H2O = 1mol water
one mol of water would contain
= 6.022 x 1023 molecules of water
For the reaction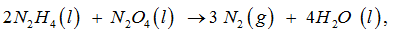
Which is the limiting reagent for this reaction ?
Chemical reactions rarely occur completely when exactly the right amounts of reactants are taken to react together to yield products as per its stoichiometry .One of the reactants will be used up ,before another runs out.Thus , the reactant which is consumed first is known as limiting reagent
For the given reaction ,
2 N2 H4 (l) + N2 O4 (l) ------> 3 N2 (g) + 4 H2 O (l)
as per its stoichiometry , if only the right and exact amount of N2 O4 is taken , the reaction may not go to completion .As such it would be consumed first & is , therefore , the limiting reagent.
Step 1
Number of moles of H2 in 67.2 L of H2
=
=3 moles
Step 2 /
Number of molecules in 1 moles of H2
=6.02 x 1023 molecules of H2
Since H2 is a diatomic gas the number of atoms in 1 mole of H2
= ( 2 x 6.02 x 1023 )
= 12.04 x 1023 atoms of H atoms
(since , one molecule of H2 contains 2 atoms ).
Number of atoms in 3 moles of H2
= ( 3 x 12.04 x 1023 )
=3.612x 1024 atoms of H.
Chemistry does not deal in explaining superconductivity.
*In superconducting materials the charecteristics of superconductivity appear when the temperature is lowered below a critical temperature.
*The onset of superconductivity is accompanied by abrupt changes in physical properties which are more related to phase transitions of the material .
These aspects of studies in properties of materials are better related to studies in the fields of Physics , eventhough principles involved in Chemistry & Physics go hand to hand.
Since, density
= mass / volume ;
and SI unit of mass is kg and that of volume is
using these SI units the unit of density is derived through dimensional analysis :
density = mass / volume
= kg / m3
or, = kg m-3
Molecular formula is the exact no. of atoms present in a molecule of a compound.
Molecular formula of a compound is related with its empirical formula as ,
Mollecular formula = ( Empirical formula ) n
where n represents a positive integer .
Since , 1L
= 1000mL
or , 1mL
=.
0.05mL
=
= 0.00005 L .
One mole is the amount of a substance that contains as many particles or entities as there are atoms in exactly 12 g of the isotope .
A mole of a substance always contains the same number of entities , no matter what the substance may be. In order to determine this number precisely , the mass of carbon -12 atom was determined by mass spectrometer and found to be equal to 1.992648 x 10-23 g. Knowing that one mole of carbon weighs 12 g . the number of atoms in it is equal to :
6.0221367 x 1023
