Some Basic Concepts of Chemistry - Online Test
Molecular mass of Glucose is calculated using the relation :
Molecular mass
= ( 6*atomic mass of C , 12* atomic mass of H , 6* atomic mass of O ) u
substituting the respective atomic masses we get ,
Molecular mass of glucose ( C6 H12 O6 )
= [ 6(12.0107)+12(1.008)+6(15.9994) ]
= 180.162 u.
Consider the chemical equation ,
----- KOH + ----- H3PO4 → ----- K3PO4 + ----- H2O.
The equation can be balanced by inserting the following in blank spaces
The given equation gets balanced by inserting the coefficients (3 , 1 ,1 ,3 ) in blank spaces from LHS to RHS . Thus, the balanced equation for the given reacion is ,
3KOH + H3PO4 → K3PO4 + 3H2O.
Consider the chemical reaction gven as ,
-----CO (g) + ----- (g) → ----- (l) + ----- .
This equation can be balanced by inserting the following in blank spaces
The balanced equation for the given equation is ,
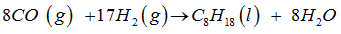
hence , the coefficients ( 8 ,17 , 1, 8 ) are inserted on blank spaces ( from LHS to RHS )
Consider the unbalanced chemical equation ,
----- Al + ------ → ------ .
The equation for above reaction can be balanced by inserting the following in blank spaces
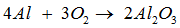
------ +----- → ------ Ba + ------ .
The above equation can be balanced by inserting the following in blank spaces from LHS to RHS respectively.
The given equation gets stoichiometrically balanced when the coefficients ( 1 , 6 , 3 , 2 ) starting from LHS to RHS are inserted in given blank spaces .
Thus , the balanced equation for the reaction is ,
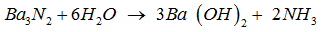
The gram molar mass of is
The gram molar mass of Ca CO3 is calculated by ,
(i) adding up the atomic masses of Ca , C & 3 O atoms & ,
(ii) representing the molar mass in grams.
Thus , gram molar mass of Ca C O3
= [ atomic mass of Ca , atomic mass of C , 3 x atomic mass of O ]
=
= ( 40 + 12 + 48 ) g
=100g
It should be noted that ,
atomic mass of Ca
= 12
atomic mass of C
= 12
atomic mass of O
= 16
Molar mass of CO2 = (atomic mass of C , 2*atomic mass of O )
= [12+2(16)] u
Since , gram molar mass
= Molar mass expressed in gms .
gram molar mass of CO2
= 44 g
