Redox Reactions - Online Test
Q1. Using the standard electrode potential, find out the pair between which redox reactions is not feasible.
Answer : Option D
Explaination / Solution:
As for this couple is negative so this couple is not possible.
Q2. An oxidation number of +2 is found in all their compounds of one of the below given options
Answer : Option D
Explaination / Solution:
Alkaline earth metals have in common an outer s- electron shell which is full; that is, that is why orbital contains its full complement of two electrons, which these elements readily lose to form cations with charge +2, and an oxidation state (oxidation number) of +2.
Q3. The oxidizing power of halogens increase in the order of
Answer : Option C
Explaination / Solution:
Halogens have high electronegativity and electron affinity. They have greater tendency to accept electrons or easily reduced, therefore they are strong oxidizing agent.
As the reduction potential decrease down the group,the oxidizing power decrease down the group the order of the oxidizing power will be as under
F2 > Cl2> Br2> I2
¥ The oxidizing power depends on,
Heat of dissociation of halogen molecule.
Electron affinity of atom.
Hydration energy of the ion.
Heat of vaporization
If a halogen has low energy of dissociation, a high electron affinity, and higher hydration of its ion, it will have high oxidizing power.
F has although low electron affinity than Cl but low dissociation energy and have high hydration energy of its ion, therefore Fluorine is strongest oxidizing agent.
Q4. In which of the following compounds, an element exhibits two different oxidation states.
Answer : Option B
Explaination / Solution:
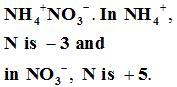
Q5. Consider the elements: Cs, Ne, I and F. Identify the element which exhibits neither the negative nor does the positive oxidation state.
Answer : Option B
Explaination / Solution:
Ne is a noble gas, so does not show any positive or negative oxidation state.
Q6. Which of the following elements does not show disproportionation tendency?
Answer : Option C
Explaination / Solution:
F (Fluorine) is most electronegative element so it always show -1 oxidation state.
Q7. Consider the elements: Cs, Ne, I and F. Identify the element(s) that exhibits only Positive oxidation state
Answer : Option C
Explaination / Solution:
Cs is alkali metal so will show positive oxidation state of +1.
Q8. The exhibition of various oxidation states by an element is also related to the outer orbital electronic configuration of its atom. Atom(s) having which of the following outermost electronic configurations will exhibit more than one oxidation state in its compounds.
Answer : Option C
Explaination / Solution:
(3d24s2) is the configuration of transition element which shows variable oxidation state.
Q9. The decomposition of hydrogen peroxide to form water and oxygen is an example of
Answer : Option C
Explaination / Solution:
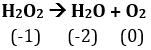
Oxidation number of O in is -1 and it is decreasing to -2 to and increases to 0 in
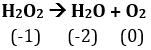
Oxidation number of O in is -1 and it is decreasing to -2 to and increases to 0 in
Q10. The electron releasing tendency of the metals, zinc, copper and silver is in the order:
Answer : Option A
Explaination / Solution:
The electron releasing tendency of the metals, zinc, copper and silver is in the order Zn>Cu>Ag
This is according to the metal activity series or electrochemical series.
