Organic compounds containing Nitrogen - Online Test
Q1. Which of the following amines can be resolved into enantiomers?
Answer : Option D
Explaination / Solution:
2-pentamine will have a chiral center.
Q2. Which of the following is a secondary amine?
Answer : Option C
Explaination / Solution:
N of this amine is bonded to two carbon atoms, therfore it is a secondary amine.
Q3. When pentanal reacts with ethylamine under conditions of acid catalysis, the major organic product is __________.
Answer : Option C
Explaination / Solution:
Imines are typically prepared by the condensation of primary amines and aldehydes and less commonly ketones:
- RNH2 + R'C(O)R → RN=C(R')(R) + H2O
Q4. Amines are soluble in
Answer : Option C
Explaination / Solution:
Amines are soluble in water due to hydrogen bonding with water.
Q5. Arrange the following compounds in order of increasing boiling point:
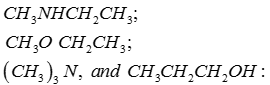
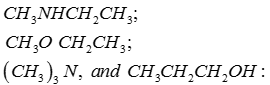
Answer : Option C
Explaination / Solution:
This is on the basis of inter molecular interactions.
Q6. When a 1° amine reacts with an alkyl sulfonyl chloride, the major organic product is __________.
Answer : Option D
Explaination / Solution:
Sulphonyl chloride reacts with primary amine to form sulphonamide.
RNH2+RI SO2Cl → RI SO2NHR + HCl
Q7. Which of the following is a tertiary amine?
Answer : Option D
Explaination / Solution:
this is tertiary amine.
Q8. When pyridine is treated with a mixture of nitric and sulfuric acids, the major product is:
Answer : Option B
Explaination / Solution:
In pyridine EAS take splace at 3 position.
Q9. Arrange the following in order of increasing basicity: aniline, p – nitroaniline, p – toluidine,and p – methoxyaniline
Answer : Option C
Explaination / Solution:
-OMe group at para position will increase the basicity more than-CH3 group at para position. While presence of –NO2at para position will decrease the basicity.
Q10. Pyridine typically undergoes________ electrophilic aromatic substitution as compared to benzene,
Answer : Option D
Explaination / Solution:
Pyridine is less reactive than benzene towards electrophilic aromatic substitution, because nitrogen is more electronegative than carbon and acts like an electron withdrawing group.
