Organic compounds containing Nitrogen - Online Test
Q1. Hinsberg’s reagent reacts with primary and secondary amines to form sulphonamides. This reagent is also known as
Answer : Option C
Explaination / Solution:
this is benzenesulphonyl chloride or hinsberg reagent.
Q2. p – toluenesulphonyl chloride does not react with
Answer : Option C
Explaination / Solution:
does not react with all these amines
Q3. p – toluenesulphonyl chloride is used to
Answer : Option C
Explaination / Solution:
p – toluenesulphonyl chloride can also be used to distinguish between , and amines in replacement of benzenesulphonylchloride.
Q4. Carbylamine reaction is one of the following in the presence of heat
Answer : Option D
Explaination / Solution:
In carbylamines reaction primary amine reacts with chloroform and base to form isocyanide having foul smell.
Q5.
Arrange the following in decreasing order of their basic strength :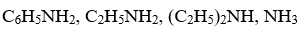
Answer : Option B
Explaination / Solution:
Alkyl amines and ammonia are more basic than aniline because in aniline lone pair on N is in resonace with benzene ring so is less available.
Also more 20 alkyl amines are more basic than primary amine and ammonia due to +I effect of the alkyl group which increases basicity.
Q6. The nitration reaction of amines can be controlled in order to obtain the desired nitro derivative as the major product by:
Answer : Option D
Explaination / Solution:
The activating power of aniline can be controlled by acetylation reaction to get desired product.
Q7. Which of the following respond to the isocyanide test?
Answer : Option B
Explaination / Solution:
Only primary amines undergo carbylamines reaction (isocyanide test)
Q8. One of the following behave like Lewis base
Answer : Option C
Explaination / Solution:
Amines behave as lewis base because of presence of lone pair on N, whereas in amides and cyanides the lone pair on nitrogen is involved in resonance. In esters, the lone pair on oxygen is involved in resonance.
Q9. The geometry of amines is
Answer : Option D
Explaination / Solution:
An amine molecule has the shape of a somewhat flattened triangular pyramid, with the nitrogen atom at the apex. An unshared electron pair is localized above the nitrogen atom.
Q10. Which of the following is a tertiary amine?
Answer : Option D
Explaination / Solution:
Benadryl have tertiary amine.
