Kinetic Theory - Online Test
Molar volume is the volume occupied by 1 mol of any (ideal) gas at standard temperature and pressure (STP : 1 atmospheric pressure, 0 C). The value of Molar volume is
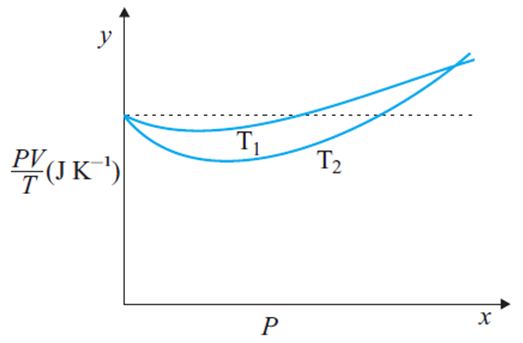
for 1 mole of ideal gas, according to ideal gas equation
hence graph must be with zero slope. So that dotted line show ‘ideal’ gas behavior and curved line shows deviation from ‘ideal’ gas behavior
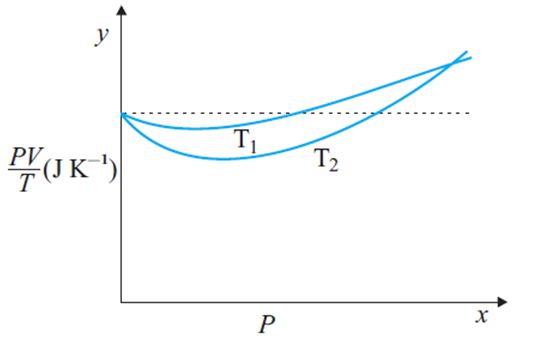
for 1 mole of ideal gas, according to ideal gas equation
hence graph must be with zero slope. So that dotted line show ‘ideal’ gas behavior and curved line shows deviation from ‘ideal’ gas behavior
A real gas behave as ideal gas at high temperature. Temperature T1 is close to dotted line.
so that >
Estimate the total number of air molecules (inclusive of oxygen, nitrogen, water vapor and other constituents) in a room of capacity 25.0 at a temperature of 27 C and 1 atm pressure.
k = boltzman constant
k = boltzman constant
k = boltzman constant
Three vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains neon (monatomic), the second contains chlorine (diatomic), and the third contains uranium hexafluoride (polyatomic). In which case is the largest?
Neon has smaller molecular wt in comparision to chlorine and uranium hexafluoride. So that is largest for neon.
At what temperature is the root mean square speed of an atom in an argon gas cylinder equal to the rms speed of a helium gas atom at – 20 ? (Atomic mass of Ar = 39.9 u, of He = 4.0 u).
