Hydrogen - Online Test
The peroxide that gives H2O2 on treatment with dilute is
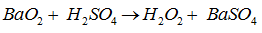
Volume strength = 11.2 x M
So M == 0.446
Strength (g/L ) = 0.446 x 34 = 15.17 g/L
The water gas shift reaction converts carbon monoxide and water to carbon dioxide and hydrogen. The reaction is catalysed by a number of different base metal catalysts, depending on the operating temperature and levels of poisons in the feedstock.
In auto-protolysis a proton is transferred between two identical molecules, one of which acts as a Bronsted acid, releasing a proton which is accepted by the other molecule acting as a Bronsted base.
Auto-protolysis is also called the self-ionization. Auto-protolysis of water is a reaction in which self-ionization of water molecules takes place to produce hydronium ion (H3O+) and hydroxide ion (OH-).
Hydrogen peraoxide is also prepared by the reaction of acid sodium peroxides. Reaction is as follow :
