Equilibrium - Online Test
Q1. At a particular temperature and atmospheric pressure, the solid and liquid phases of a pure substance can exist in equilibrium. Which of the following term defines this temperature?
Answer : Option B
Explaination / Solution:
Normal melting both the phases liquid and solid exist in equilibrium.
Q2. The aqueous solution of sugar does not conduct electricity. However, when sodium chloride is added to water, it conducts electricity. How will you explain this statement on the basis of ionisation and how is it affected by concentration of sodium chloride?
Answer : Option A
Explaination / Solution:
NaCl ionises completely in water and increase in concentration of salt increases conductance.
Q3. does not have proton but still acts as an acid and reacts with NH3. Why is it so? What type of bond is formed between the two?
Answer : Option B
Explaination / Solution:
is a Lewis acid as B has its vacant p orbital and coordinate bond is formed as donates its lp of electrons to the vacant orbital of B.
Q4. is dissolved in water to give 500 mL of solution. The pH of the solution is
Answer : Option C
Explaination / Solution:
pOH=-log[] and here [OH-] = (0.3 x 1000)/(74 x500) While pH=14-pOH
Q5. Calculate the hydrogen ion concentration in the human blood whose pH is 7.38.
Answer : Option A
Explaination / Solution:
pH=-log[]
Q6.
The ionization constant of acetic acid is . Calculate the degree of dissociation of acetic acid in its 0.05 M solution. Calculate the concentration of acetate ion in the solution and its pH.
Answer : Option B
Explaination / Solution:
Ka = and pH=-log(Cα).
Q7. It has been found that the pH of a 0.01M solution of an organic acid is 4.15.Calculate the concentration of the anion, the ionization constant of the acid and its
Answer : Option D
Explaination / Solution:
Ka = and pH=-log(Cα) and pKa=-logKa
Q8. 0.023 g of sodium metal is reacted with of water. The pH of the resulting solution is ______.
Answer : Option A
Explaination / Solution:
Moles of NaOH formed = 0.023/23 =0.001 Conc of NaOH=0.001x1000/100 =0.01 pOH=-log[OH-] =2 pH=14-2=12
Q9.
A vessel at 1000 K contains with a pressure of 0.5 atm. Some of the is converted into CO on the addition of graphite. If the total pressure at equilibrium is 0.8 atm, the value of K is
Answer : Option A
Explaination / Solution:
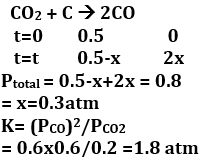
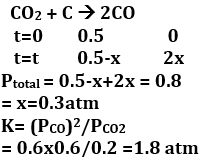
Q10. What is the best description of the change that occurs when is dissolved in water?
Answer : Option C
Explaination / Solution:
Oxide ion donates a pair of electrons
