Equilibrium - Online Test
Q1. At a given temperature equilibrium is possible only in a system which is a
Answer : Option D
Explaination / Solution:
Equilibrium is possible only in a closed system at a given temperature for physical processes. When equilibrium is attained for a physical process, it is characterized by constant value of one of its parameters at a given temperature.
Q2. For liquid ⇌⇌ vapour equilibrium, at a given temperature, the constant is
Answer : Option B
Explaination / Solution:
The vapour pressure is constant at a given temperature for liquid ⇌ vapour equilibrium. The equilibrium vapour pressure of a compound at a given temperature is an intrinsic property of the compound, meaning it does not depend on variables like volume, quantity of the compound, etc.
Q3. The aqueous solution of sugar does not conduct electricity. However, when sodium chloride is added to water, it conducts electricity. How will you explain this statement on the basis of ionisation and how is it affected by concentration of sodium chloride?
Answer : Option B
Explaination / Solution:
Sugar being a non-electroyte, does not ionize in water, whereas NaCl ionizes completely in water and produce Na+ and Cl- ions, which help in the conduction of electricity and when concentration of NaCl is increased , more Na+ and Cl- ions will be produced, hence conductance increases.
Q4. For dissolution of solids in liquids, at a given temperature, the constant is
Answer : Option D
Explaination / Solution:
This is considered at equilibrium process.
Q5. For dissolution of gases in liquids, the concentration of a gas in liquid is.
Answer : Option C
Explaination / Solution:
For dissolution of gases in liquids, the concentration of a gas in liquid is proportional to the pressure of the gas over the liquid
Q6. the concentrations in an equilibrium mixture are related by the following equilibrium equation, Where Kc is called as
Answer : Option B
Explaination / Solution:


Q7. For reactions involving gases, however, it is usually more convenient to express the equilibrium constant in terms of
Answer : Option C
Explaination / Solution:
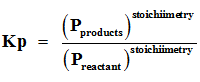
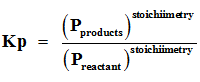
Q8. If the Kc is neither too small nor too large, one of the following will happen
Answer : Option C
Explaination / Solution:
Rxn is at equilibrium.
Q9. A particular ratio of product to reactant helps in predicting the direction in which a given reaction will proceed at any stage. is called
Answer : Option C
Explaination / Solution:
Reaction Quoitennt (Q) =K ( equilibrium) Q>K (backward Reaction) Q
Q10. The value of for a reaction is directly related to the thermodynamics of the reaction and in particular,
Answer : Option A
Explaination / Solution:
Δ G = -RTlnK
