Electrochemistry - Online Test
Q1. Three electrolytic cells A,B,C containing solutions of ZnSO4, AgNO3 and CuSO4, respectively are connected in series. A steady current of 1.5 amperes was passed through them until 1.45 g of silver deposited at the cathode of cell. How long did the current flow? What mass of copper and zinc were deposited?
Answer : Option C
Explaination / Solution:
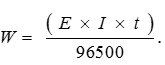
And mass of Zn and Cu deposited will be in ratio of their equivalent mass.
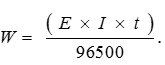
And mass of Zn and Cu deposited will be in ratio of their equivalent mass.
Q2. Resistance of 0.2 M solution of an electrolyte is 50 Ω. The specific conductance of the solution is 1.3 S . If resistance of the 0.4 M solution of the same electrolyte is 260 Ω, its molar conductivity is
Answer : Option C
Explaination / Solution:
κ= G × cell constant and G= 1/R.
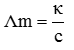
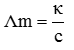
Q3. The standard emf of galvanic cell involving 3 moles of electrons in its redox reaction is 0.59 V. The equilibrium constant for the reaction of the cell is
Answer : Option A
Explaination / Solution:
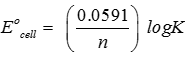
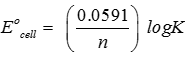
Q4. An increase in equivalent conductance of a strong electrolyte with dilution is mainly due to
Answer : Option C
Explaination / Solution:
Equivalent conductance increases on dilution for a strong electrolyte because of increase in mobility of ions.
Q5.
For the reduction of silver ions with copper metal the standard cell potential was found to be +0.46V at 25°C. The value of standard Gibbs energy, ΔG° wll be (F = 96500 C )
Answer : Option A
Explaination / Solution:
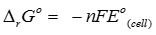
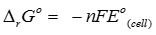
Q6. Which of the following electrolytic solutions has the least specific conductance?
Answer : Option D
Explaination / Solution:
Specific conductance decreases with dilution because of decrease in the number of ions per unit volume.
Q7. The highest electrical conductivity of the following aqueous solutions is of
Answer : Option C
Explaination / Solution:
Acidity increases on attaching electron withdrawing group because of stability of conjugate base.
Q8. Saturated solution of is used to make 'salt bridge' because
Answer : Option C
Explaination / Solution:
Saturated solution of is used in salt bridge because of same mobility o f and
Q9. Li occupies higher position in the electrochemical series of metals as compared to Cu since
Answer : Option A
Explaination / Solution:
Li is alkali metal /Li = -3.045V.``
Q10. Hydrogen gas is not liberated when the following metal is added to dil. HCl.
Answer : Option C
Explaination / Solution:
Ag is less reactive than Hydrogen.
