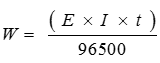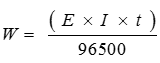Electrochemistry - Online Test
Q1. The conductivity of 0.20 M solution of KCl at 298 K is 0.0248 S . Calculate its molar conductivity.
Answer : Option C
Explaination / Solution:
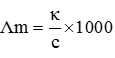
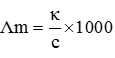
Q2. The resistance of a conductivity cell containing 0.001M KCl solution at 298 K is 1500 Ω. What is the cell constant if conductivity of 0.001M KCl solution at 298 K is 0.146 × 10–3 S .
Answer : Option D
Explaination / Solution:
Κ= G x cell constant and G= 1/R.
Q3. Conductivity of 0.00241 M acetic acid is 7.896 × S . If for acetic acid is 390.5 S , what is its dissociation constant?
Answer : Option D
Explaination / Solution:
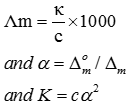
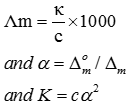
Q4. How much charge is required for the reduction of 1 mol of to Al?
Answer : Option B
Explaination / Solution:
For reduction of 1 mol of to Al , 3 mol of electrons are required so total charge will be 3F.
Q5. How much charge is required for the reduction of 1 mol of to Cu?
Answer : Option C
Explaination / Solution:
For reduction of 1 mol of to Cu, 2 mol of electrons are required so total charge will be 2F.
Q6. How much electricity in terms of Faraday is required to produce 20.0 g of Ca from molten CaCl2?
Answer : Option B
Explaination / Solution:
20g is 0.5 mol of and for 1mol 2F charge is required.
Q7. How much electricity in terms of Faraday is required to produce 40.0 g of Al from molten ?
Answer : Option C
Explaination / Solution:
Moles of Al= 40/27. For 1 mol of Al deposition 3F charge is required.
Q8. How much electricity is required in coulomb for the oxidation of 1 mol of to ?
Answer : Option C
Explaination / Solution:
For 1 mol to 2 mol of electrons are required.
Q9.
How much electricity is required in coulomb for the oxidation of 1 mol of FeO to ?
Answer : Option C
Explaination / Solution:
For converting FeO to 1mol of electrons are required.
Q10.
A solution of Ni()2 is electrolyzed between platinum electrodes using a current of 5 amperes for 20 minutes. What mass of Ni is deposited at the cathode?
Answer : Option D
Explaination / Solution: