Chemical Bonding and Molecular Structure - Online Test
Q1. The state of hybridization of the central atom and the number of lone pairs over the central atom in are
Answer : Option C
Explaination / Solution:
CN= 0.5[V+M-C+A]
For POCl3 V=5 M=3 C =A=0 So CN=4
so hybridization is sp3. And there are 4 bps so 0 lps.
Q2. When is converted into
Answer : Option A
Explaination / Solution:
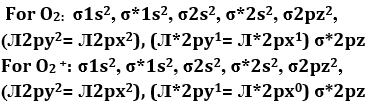
Bond order =
For O2 = = 2
For O2+ = = 2.5
Q3. Which one of the following conversions involve change in both hybridization and shape?
Answer : Option C
Explaination / Solution:
BF3 has sp2 hybridisation and its shape is triangular planar While BF4- has sp3 hybridisation and shape is tetrahedral.
Q4.
Based on VSEPR theory, the number of 90 degree F — Br — F angles in is
Answer : Option D
Explaination / Solution:
Steric no. of BrF5 is 6 so geometry is octahedral. It will have 5 bps and one lp which is present on axial position. Because of distortion caused by lp there is no 90o F—Br—F bond angle in BrF5 .
Q5. The species having pyramidal shape is
Answer : Option B
Explaination / Solution:
SF2O has pyramidal shape as it is sp3 hybridised and it has 3bp and 1 lp.
Q6. For a stable molecule the value of bond order must be
Answer : Option B
Explaination / Solution:
Bond order∝Stability
Hence, for a stable molecule the value of bond order must be positive. When bond order is zero and negative the molecule will not exist.
Q7. In acetylene molecule, between the carbon atoms there are
Answer : Option D
Explaination / Solution:
acetylene is C2H2.
C atoms are bound with triple bond i.e. 1 sigma and 2 pie bonds.
Q8. In ion the formal charge on the oxygen atom of P–O bond is
Answer : Option D
Explaination / Solution:
Formal charge on the oxygen atom of P–O bond is – 1
Q9. In which of the following substances will hydrogen bond be strongest?
Answer : Option A
Explaination / Solution:
Out of all H2O will form strongest hydrogen bonding due to high electronegativity of O.
Q10. When a gas phase atom in its ground state gains an electron. This is called
Answer : Option D
Explaination / Solution:
electron gain enthalpy is the energy released when an electron is gained by isolated gaseous atom to form isolated gaseous anion.
