Chemical Bonding and Molecular Structure - Online Test
Q1. in case of a heteronuclear molecule like HF, the shared electron pair between the two atoms gets displaced more towards fluorine since the electronegativity of fluorine is far greater than that of hydrogen. This results in the molecul
Answer : Option C
Explaination / Solution:
because of slight shifting of electron towards more electronegative element like F in HF brings a small amount of negative charge on F and positive charge on H which results in dipole moment.
Q2. Elements in which apart from 3s and 3p orbitals, 3d orbitals also available for bonding In a number of compounds of these elements there are more than eight valence electrons around the central atom. One such example is
Answer : Option D
Explaination / Solution:
In , S has more than 8 electrons in valence shell.It has 12 electrons in its valence shell.
Q3. The shift in electron density is symbolised by crossed arrow in the below diagram. It depicts
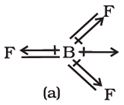

Answer : Option B
Explaination / Solution:
The cross base arrow demonstrates the net dipole. On the cross-base arrow, the cross represents the positive charge and the arrow represents the negative charge.
Q4.
In , molecule below the dipole moment is zero although the B- F bonds are oriented at an angle of to one another. Net dipole moment in molecule is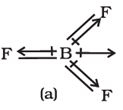
Answer : Option A
Explaination / Solution:
BF3 has symmetrical structure in which the three B-F bonds are oriented at an angle of 120° to one another. Also the three bonds lie in one plane and the dipole moments of these bonds cancel one another giving net dipole moment equal to zero.
Q5. The shape of the below molecule is
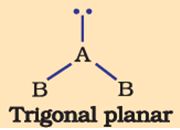

Answer : Option C
Explaination / Solution:
Bent shape is the shape of a molecule with 3 atoms that have a bond angle less than 180°.
Q6. Polarity in a molecule and hence the dipole moment depends primarily on electronegativity of the constituent atoms and shape of a molecule. Which of the following has the highest dipole moment?
Answer : Option C
Explaination / Solution:
CO2 is linear, so even though the C−O bonds have individual dipole moments, the overall dipole moment is zero as these cancel out (they point in opposite directions, as shown in the diagram below).
On the other hand, H2O is "bent", which means that the individual dipole moments of the bond are at an angle to each other and because of Presence of lp on head of O atom in H2O therefore it has max dipole moment.
Q7. Ionic crystalline compounds formed by ion-formation by electron transfer proves
Answer : Option D
Explaination / Solution:
Ionic crystalline compounds formed by ion-formation by electron transfer proves electronic theory of chemical bonding.
Q8. The tetrahedral molecule below where there are two lone pairs of electrons is
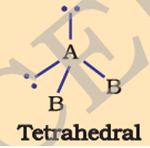

Answer : Option B
Explaination / Solution:
An important feature of water is its polar nature. The structure has a bent molecular geometry for the two hydrogens from the oxygen vertex. The oxygen atom also has two lone pairs of electrons.
Q9. The criterion of overlap of atomic orbitals is the main factor for the formation of
Answer : Option D
Explaination / Solution:
The valence bond theory is introduced to describe bonding in organic molecules. In this model, bonds are considered to form from the overlapping of two atomic orbitals on different atoms, each orbital containing a single electron.
In general, greater the overlap, stronger is the bond formed between the two atoms. Thus, according to the orbital overlap concept, atoms combine together by overlapping their orbital and thus forming a lower energy state where their valence electrons with opposite spin, pair up to form covalent bond.
Q10. Octet of electrons, represents a particularly stable electronic arrangement. Atoms achieve the stable octet when they are linked by chemical bonds. This rule is associated with one of the following theories
Answer : Option D
Explaination / Solution:
G.N.Lewis, proposed the octet rule to explain the valence electron sharing between atoms that resulted in a bonding type with the atoms attaining noble gas electronic configuration.
The statement is : "a bond is formed between two atoms by mutual sharing of pairs of electrons to attain a stable outer-octet of electrons for each atom involved in bonding". This type of valence electron sharing between atoms is termed as covalent bonding.
