Chemistry - Online Test
Q1. The
amount of heat exchanged with the surrounding at constant temperature and
pressure is given by the quantity
Answer : Option B
Explaination / Solution:
No Explaination.
Q2. The following compounds are called
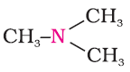

Answer : Option D
Explaination / Solution:
The following compound is tertiary amine.
Q3. When the rate of evaporation is equal to the rate of condensation, it is called
Answer : Option D
Explaination / Solution:
At equilibrium, the rate of evaporation is equal to the rate of condensation.
It may be represented by H2O (l) ⇌ H2O (vap)
The double half arrows indicate that the processes in both the directions are going on simultaneously. The mixture of reactants and products in the equilibrium state is called an equilibrium mixture
Q4. The molality of a solution containing 1.8g of glucose dissolved in 250g of water is
Answer : Option D
Explaination / Solution:
molality = number of moles of solute / weight of solvent (in
kg)
= (1.8/180) / 0.25 = 0.01/0.25 = 0.04M
Q5. Physical behavior of the states differs greatly even though chemical behavior of the three states is identical because
Answer : Option C
Explaination / Solution:
chemical properties depend on intramolecular bonding forces whereas physical properties depend on intermolecular nonbonding forces
Q6. Concentration
of the Ag+ ions in a saturated solution of Ag2 C2O4
is 2.24 ×10-4mol L-1
solubility product of Ag2 C2O4 is
Answer : Option D
Explaination / Solution:
Ag2C2O4
↔ 2Ag+
+ C2O42-
[ Ag+ ] = 2.24 × 10-4
mol L-1
[ C2 O42-
] = { 2.24×10-4 }/2 = mol L-1
= 1.12×10-4 mol L-1
Ksp = [Ag+]2[C2O42-]
= (2.24×10-4 mol-4 L-1
)2 (1.12×10-4 mol L-1)
= 5.619×10-12mol3
L-3
Q7. Glycerol is
Answer : Option C
Explaination / Solution:
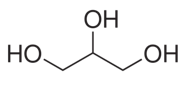
Glycerol and trihydroxypropane are common names. The IUPAC name is propane-1,2,3-triol. The common name glycerol comes from the root glyco- which means sweet. Glucose, glycogen, and glycerin (another name for glycerol) have the same etymology.

Glycerol and trihydroxypropane are common names. The IUPAC name is propane-1,2,3-triol. The common name glycerol comes from the root glyco- which means sweet. Glucose, glycogen, and glycerin (another name for glycerol) have the same etymology.
Q8. Bauxite has the composition
Answer : Option B
Explaination / Solution:
No Explaination.
Q9. Which of these fuels given below is not a hydrocarbon?
Answer : Option B
Explaination / Solution:
Because hydrocarbons contain both C and H and hence Hydrogen is not a hydrocarbon. .
Q10. Example of solution of liquid in gas is
Answer : Option A
Explaination / Solution:
Solute is water (liquid) and solvent is gas( air).
