Chemistry - Online Test
in hybridization as % of 's'character increases electronegativity also increases.
more the s charater, more will be the electronegativity of that atom.
increasing order ofelectronegativity with %s character is sp (50%) > sp2(33.33%) > sp3(25%)
Potassium is lighter than sodium (Refer table 5.3)
The correct order of density is Li < K < Na
< Rb < Cs
Oxidation means addition of oxygen, removal of hydrogen, increase in oxidation number, addition of a electronegative element or removal of an electropositive element
The term oxidation was first used to describe chemical reactions in which oxygen was added to an element of a compound. Later on the term oxidation was extended to describe many more reactions which occurred without the use of even oxygen.
Addition of Oxygen: Oxidation is a chemical reaction in which oxygen is added to any chemical species (atom, ion or molecule). Example
2Mg + O2 → 2MgO
S + O2 → SO2
2CO + O2 → 2CO2
Na2SO3 + H2O2 → Na2SO4 + H2O
Removal of Hydrogen : Oxidation is a chemical reaction in which hydrogen is removed from any chemical species (atom, ion or molecule).
H2S + Cl2 → 2HCl + S
4HI + O2 → 2H2O + 2I2
4HI + MnO2 → MnCl2 + 2H2O + Cl2
Addition of an Electronegative Element: Oxidation is a chemical reaction in which an electronegative element is added into any chemical species (atom, ion or molecule).
Fe+ S → FeS (Oxidation of iron)
SnCl2 + Cl2→ SnCl4(Oxidation of stannous chloride)
2Fe + 3F2 → 2FeF3(Oxidation of iron)
Removal of an Electropositive Element: Oxidation is a chemical reaction in which an electropositive element is removed from any chemical species (atom, ion or molecule).
2KI + H2O2 → 2KOH + I2 (Oxidation of potassium iodide)
2K2MnO4 + Cl2 → 2KCl + 2KMnO4 (Oxidation of potassium manganate)
2KI + Cl2 → 2KCl + I2 (Oxidation of potassium iodide)
A substance which brings oxidation is known as oxidizing agentt
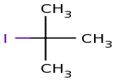 actually
here firstly we select longest chain and it is of 3 carbon.and at
number 2 position there is iodo as well as methyl ,so its name is 2-iodo
2-methyl propane.
actually
here firstly we select longest chain and it is of 3 carbon.and at
number 2 position there is iodo as well as methyl ,so its name is 2-iodo
2-methyl propane.is 2 – Iodo 2·methyl propane
Kb = 0.8 × 10–5
Kf = 1.6 × 10–4
Keq = Kf / Kb = 1.6X10-4
/ 0.8X10-5 = 20
